Denise R. Aberle
Vision-Language Model-Based Semantic-Guided Imaging Biomarker for Early Lung Cancer Detection
Apr 30, 2025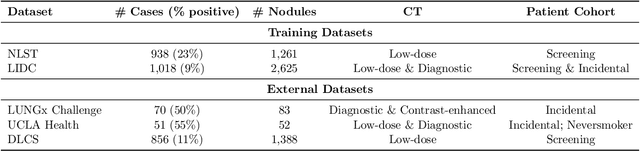
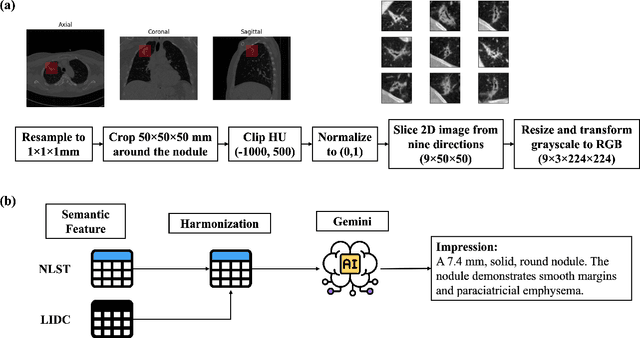
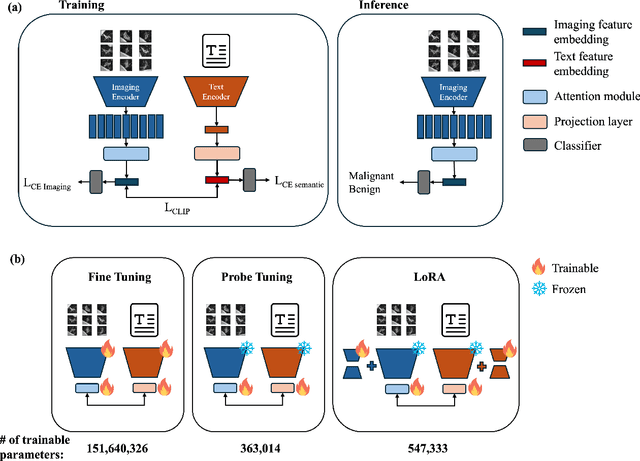
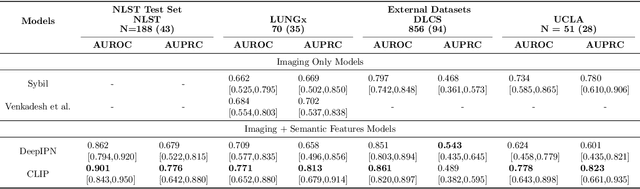
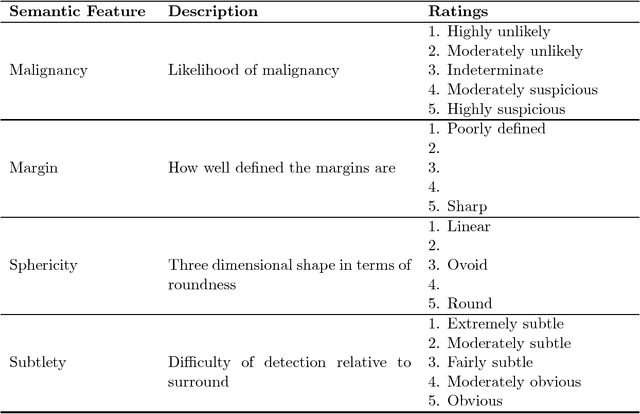
Abstract:Objective: A number of machine learning models have utilized semantic features, deep features, or both to assess lung nodule malignancy. However, their reliance on manual annotation during inference, limited interpretability, and sensitivity to imaging variations hinder their application in real-world clinical settings. Thus, this research aims to integrate semantic features derived from radiologists' assessments of nodules, allowing the model to learn clinically relevant, robust, and explainable features for predicting lung cancer. Methods: We obtained 938 low-dose CT scans from the National Lung Screening Trial with 1,246 nodules and semantic features. The Lung Image Database Consortium dataset contains 1,018 CT scans, with 2,625 lesions annotated for nodule characteristics. Three external datasets were obtained from UCLA Health, the LUNGx Challenge, and the Duke Lung Cancer Screening. We finetuned a pretrained Contrastive Language-Image Pretraining model with a parameter-efficient fine-tuning approach to align imaging and semantic features and predict the one-year lung cancer diagnosis. Results: We evaluated the performance of the one-year diagnosis of lung cancer with AUROC and AUPRC and compared it to three state-of-the-art models. Our model demonstrated an AUROC of 0.90 and AUPRC of 0.78, outperforming baseline state-of-the-art models on external datasets. Using CLIP, we also obtained predictions on semantic features, such as nodule margin (AUROC: 0.81), nodule consistency (0.81), and pleural attachment (0.84), that can be used to explain model predictions. Conclusion: Our approach accurately classifies lung nodules as benign or malignant, providing explainable outputs, aiding clinicians in comprehending the underlying meaning of model predictions. This approach also prevents the model from learning shortcuts and generalizes across clinical settings.
Advancing Precision Oncology Through Modeling of Longitudinal and Multimodal Data
Feb 11, 2025



Abstract:Cancer evolves continuously over time through a complex interplay of genetic, epigenetic, microenvironmental, and phenotypic changes. This dynamic behavior drives uncontrolled cell growth, metastasis, immune evasion, and therapy resistance, posing challenges for effective monitoring and treatment. However, today's data-driven research in oncology has primarily focused on cross-sectional analysis using data from a single modality, limiting the ability to fully characterize and interpret the disease's dynamic heterogeneity. Advances in multiscale data collection and computational methods now enable the discovery of longitudinal multimodal biomarkers for precision oncology. Longitudinal data reveal patterns of disease progression and treatment response that are not evident from single-timepoint data, enabling timely abnormality detection and dynamic treatment adaptation. Multimodal data integration offers complementary information from diverse sources for more precise risk assessment and targeting of cancer therapy. In this review, we survey methods of longitudinal and multimodal modeling, highlighting their synergy in providing multifaceted insights for personalized care tailored to the unique characteristics of a patient's cancer. We summarize the current challenges and future directions of longitudinal multimodal analysis in advancing precision oncology.
An Interpretable Deep Hierarchical Semantic Convolutional Neural Network for Lung Nodule Malignancy Classification
Jun 02, 2018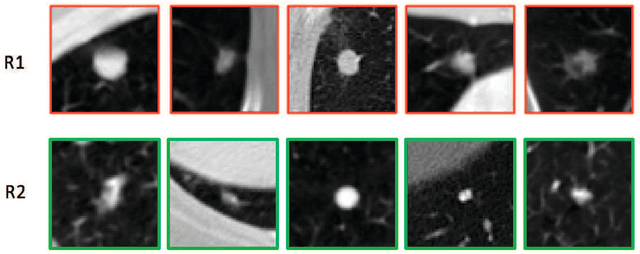


Abstract:While deep learning methods are increasingly being applied to tasks such as computer-aided diagnosis, these models are difficult to interpret, do not incorporate prior domain knowledge, and are often considered as a "black-box." The lack of model interpretability hinders them from being fully understood by target users such as radiologists. In this paper, we present a novel interpretable deep hierarchical semantic convolutional neural network (HSCNN) to predict whether a given pulmonary nodule observed on a computed tomography (CT) scan is malignant. Our network provides two levels of output: 1) low-level radiologist semantic features, and 2) a high-level malignancy prediction score. The low-level semantic outputs quantify the diagnostic features used by radiologists and serve to explain how the model interprets the images in an expert-driven manner. The information from these low-level tasks, along with the representations learned by the convolutional layers, are then combined and used to infer the high-level task of predicting nodule malignancy. This unified architecture is trained by optimizing a global loss function including both low- and high-level tasks, thereby learning all the parameters within a joint framework. Our experimental results using the Lung Image Database Consortium (LIDC) show that the proposed method not only produces interpretable lung cancer predictions but also achieves significantly better results compared to common 3D CNN approaches.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge